Opinion | What Does the Recent Pig-to-Human Kidney Transplant Mean for Tissue Therapeutics?
In a pioneering procedure, a team of surgeons at New York University Langone Health Grossman School of Medicine in New...
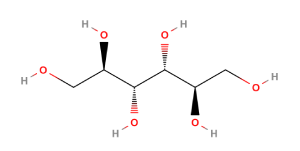
Mannitol is a hexahydric sugar alcohol (polyol) that corresponds to 1,2,3,4,5,6-Hexanehexol and the chemical formula, C6H14O6. It is closely related to Mannose and is isomeric with Sorbitol. Mannitol has a sweet taste, approximately as sweet as glucose and half as sweet as sucrose, and imparts a cooling sensation on the tongue. Pharmaceutical grades of mannitol are supplied as odourless white crystalline powders or free-flowing granules.
Pharmacopoeial Compliance: USP-NF; Ph.Eur; J.P; B.P
Synonyms and Trade Names: Mannitol; D-Mannitol; C* PharmaMannidex®; E421; Emprove® Mannitol; Mannogem®; Pearlitol®
Uses and Applications: Plasticizer; Sweetening Agent; Tablet and Capsule Diluent; Therapeutic Agent and Tonicity Agent

Mannitol (D-mannitol) is a polyol (hexahydric alcohol) chemically similar to mannose and an isomer of Sorbitol. It occurs as a white, odorless, crystalline powder, or free-flowing granules. It has a sweet taste, approximately as sweet as glucose and half as sweet as sucrose, and imparts a cooling sensation in the mouth.
Mannitol may be extracted from the dried sap of manna and other natural sources by means of hot alcohol or other selective solvents. It is commercially produced by the catalytic or electrolytic reduction of monosaccharides such as mannose and glucose.
As an isomer of sorbitol, mannitol differs from sorbitol in the planar orientation of the OH group on the second carbon atom. Each isomer is characterized by its own individual set of properties, the most important difference being the response to moisture. Sorbitol is hygroscopic, while mannitol resists moisture sorption, even at high relative humidities.
Microscopically appears as orthorhomibic needles when crystallized from alcohol. Mannitol shows polymorphism.
| Chemical Name | D-Mannitol |
| CAS Registration Number | [69-65-8] |
| Empirical Formula | C6H14O6 |
| Molecular weight | 182.17 |
| EC Number | 200-711-8 |
| UNII Code (FDA) | 3OWL53L36A |
Mannitol is an approved pharmaceutical excipient. It is listed in the USP-NF, PhEur and JP. It is also GRAS listed and accepted for use as a food additive in Europe (E421). Mannitol is also included in the FDA Inactive Ingredients Database (IP, IM, IV, and SC inect1ons; infusions; buccal, oral and sublingual tablets, powders and capsules; ophthalmic preparations; topical solutions).
| Form | Solid, powder |
| Appearance | White, crystalline powder or granules |
| Compressibility | Highly compressible |
| Bulk density | 0.40 -0.70 g/ml (typical) |
| Tapped density | 0.50 – 0.60 g/ml (Powder)
0.400 – 0.800 g/ml (Granules ) |
| pKa (18 oC) | 13.5 |
| Flash point | <150 oC |
| Flowability | Powder is cohesive while granules are free-flowing. |
| Particle size distribution | 50 – 500µm |
| Melting point | 166 – 1680C |
| Heat of combustion | 16.57 kJ/g |
| Heat of solution (25 oC) | -120.9 J/g |
| Moisture content | Non-hygroscopic |
| Osmolarity | At a concentration of 5.07 % w/v in water, Mannitol is isosmotic with plasma |
| Refractive index | 1.333 |
| Solubility | Soluble in water, ethanol, glycerine and alkalis |
| Specific surface area | 0.37 – 0.39 m2/g |
| USP-NF | Ph.Eur | J.P | |
| Official name | Mannitol | Mannitol | D-Mannitol |
| Authorised use | Excipient | Excipient
Therapeutic |
Excipient |
| Definition | specified | specified | specified |
| Identification | specified | specified | specified |
| Characters | n/a | specified | n/a |
| Appearance of solution | n/a | specified | specified |
| Melting range | 164-169 oC | 165 -170 oC | 166 -169 oC |
| Specific rotation | +137o to 145o | +23 o to +25 o | +137o to 145o |
| Conductivity | n/a | ≤ 20µ S.cm-1 | n/a |
| Acidity | specified | n/a | specified |
| Loss on drying | ≤ 0.3% | ≤ 0.5% | ≤ 0.3% |
| Chloride | ≤ 0.007% | n/a | ≤ 0.007% |
| Sulfate | ≤ 0.01% | n/a | ≤ 0.01% |
| Arsenic | ≤ 1 ppm | n/a | ≤ 1.7 ppm |
| Lead | n/a | ≤ 0.5 ppm | n/a |
| Nickel | n/a | ≤ 1 ppm | specified |
| Heavy metals | n/a | n/a | ≤ 5 ppm |
| Reducing sugars | specified | ≤ 0.2% | specified |
| Residue on ignition | n/a | n/a | ≤ 0.10% |
| Related substances | n/a | specified | n/a |
| Bacterial endotoxins | n/a | ≤ 4 IU/g | n/a |
| n/a | ≤ 2.5 IU/g | n/a | |
| Microbial contamination | n/a | ≤ 100 cfu/g | n/a |
| Assay (dried basis) | 96.0 -101.5% | 98.0 -102.0% | ≥ 98.0% |
| Labelling | n/a | n/a | specified |
Key: n/a Specification is not listed
*All claims with respect to conformity are subject to our Terms and Conditions. No express or implied warranty is made for specific properties or fitness for any particular application or purpose.
Mannitol is a pharmaceutical excipient and food additive that functions as a tablet/capsule diluent, sweetening agent, therapeutic/tonicity agent, and plasticizer. While not as popularly used as Lactose monohydrate, Calcium carbonate or Starch, Mannitol has been growing in importance in recent years, and increasingly many NPD projects look to consider it as an excipient of choice. The reasons are mainly due to its favourable tabletting properties, cost-effectiveness, inertness to active pharmaceutical ingredients, low hygroscopicity, and safety.
Mannitol is suitable for use as a filler and diluent in tablets and granules at levels of 10 – 90% w/w. It is particularly valuable owing to its low hygroscopicity, high compressibility and versatility since it is amenable to both direct compression and wet granulation processing approaches. For direct compression tabletting applications, granular and spray-dried grades are available from suppliers such as SPI Pharma (Mannogem® Mannitol) and Roquette Freres (Perlitol® Mannitol).
Granular Mannitol grades are designed to flow very well, and for the most part, these desirable properties are transferred into formulations. The only limitation is that they are very abrasive and not typically used in concentrations in excess of 25 %. Abrasiveness can be mitigated with lubricants (1% w/w Calcium stearate or 1-2% w/w Magnesium stearate).
There are several self-lubricating grades availed by a number of excipient manufacturers. These grades are typically co-processed with Starch, Glyceryl Monostearate or other excipients. An example of a self-lubricating grade is LUBRITOSE® Mannitol from Kerry Group.
Standard crystalline or powdered Mannitol grades are reserved for wet granulation owing to their poor flowability. In spite of this drawback, granulations that contain Mannitol have the advantage of being able to dry readily. Suitable binders for preparing granulations of powdered mannitol are Methylcellulose, Starch paste, Povidone, and Sorbitol.
Mannitol is also specifically selected in the formulation of antacid preparations, nutraceutical tablets, powders, as well as mouth disintegrating dosage forms (chewable, sublingual, orally disintegrating tablets (ODTs), and lozenges). It provides excellent taste and mouthfeel, which is particularly important for these types of dosage forms. Mannitol’s negative heat of solution, sweetness, and mouthfeel not only helps enhance the perception of flavours but also helps with optimising taste-masked formulations.
Mannitol also functions as an osmotic direct, a diagnostic agent for measuring kidney function or as an adjunct in the management of renal disease, and as an agent to reduce intracranial pressure (in the treatment of cerebral) oedema or reduce intraocular pressure.
Mannitol (20 -90% w/w) can be added to lyophilized formulations to help produce a stiff, homogeneous mass that improves the appearance of the lyophilized plug in a vial. A pyrogen-free grade is supplied specifically to meet this application.
Mannitol can be added to antacid suspensions of Aluminium hydroxide to prevent thickening and to permit consistent administration/use of the product.
Mannitol is also widely utilised in a variety of food products taking advantage of its sweetening properties. It is recommended for the development of sugar-free confectionery and energy-reduced products, both for its tooth-friendliness (non-cariogenic), and low caloric value.
Being a naturally occurring substance and an ordinary component of the regular human diet, Mannitol is generally considered a safe and non-toxic substance. It is used in foods as a bulking agent and non-nutritive sweetener. Following intravenous administration, Mannitol does not undergo significant metabolism and is only nominally reabsorbed by the kidneys, with about 80% of the administered dose being eliminated in the urine. However, it can produce laxative effects when consumed orally in large amounts. It is advised that when daily intake is likely to exceed 20g product label should include a warning statement regarding laxative effects.
It has been reported that Mannitol is associated with specific adverse reactions when used as a therapeutic agent (e.g 20% w/v aqueous intravenous infusion). Since the amount of Mannitol used as an excipient is significantly lower than when used therapeutically, the importance of these adverse reactions when used as an excipient is less significant. Allergic and hypersensitive-type reactions, which have been reported, are still relevant when Mannitol is used as a formulation aid.
An acceptable daily intake of mannitol has not been specified by the WHO since the amount consumed as a sweetening agent was not considered to represent a hazard to health.
Toxicology: LD50 (rat, IV): 9.69 g/kg; LD50 (rat, oral): 13.5 g/kg
Mannitol is generally a stable excipient both as dry material and when constituted into aqueous solutions. Mannitol solutions are not affected by dilute acids, alkalis, or atmospheric oxygen. Mannitol does not undergo Maillard reaction. It has good thermal stability, and solutions can be sterilized by autoclaving with no adverse physical or chemical changes.
Powdered Mannitol is assigned a shelf life of 24-36 months. The bulk material should be stored in a tightly-closed container in a cool, dry place away from direct heat or moisture.
Mannitol solutions (concentration ≥ 20% w/v) can be precipitated by Potassium chloride or Sodium chloride. Mannitol is also not compatible with Xylitol infusions.
When handling Mannitol, observe established SHEQ precautions appropriate to the circumstances and quantity of material processed. As Mannitol may be irritant to the eyes and mucous membranes, the use of appropriate PPE (eye protection and gloves) is recommended.
Mannitol is found in a wide variety of plants and seaweed, and can be directly extracted from these natural sources. As a biodegradable, natural substance, it is considered safe for the environment with no long-term impact on ecology or marine life. Mannitol excipient grade achieved a total score of 80/100 by the Excipients Forum Sustainable Chemistry Score.™
The Mannogem® line of Mannitol from SPI Pharma is the widest in the pharmaceutical industry. The company was the first to introduce Mannitol powder and Mannitol for direct compression. Click here for a guide on selection of Mannogem® grade of Mannitol here.
[3] H. Ando, S. Watanabe, T. Ohwaki, Y. Miyake, Crystallization of excipients in tablets, Journal of Pharmaceutical Sciences, 74 (1985) 128-131.
[4] S.N.C. Roberts, A.C. Williams, I.M. Grimsey, S.W. Booth, Quantitative analysis of mannitol polymorphs. FT-Raman spectroscopy, Journal of Pharmaceutical and Biomedical Analysis, 28 (2002) 1135-1147.
[5] T. Yoshinari, R.T. Forbes, P. York, Y. Kawashima, Moisture induced polymorphic transition of mannitol and its morphological transformation, International Journal of Pharmaceutics, 247 (2002) 69-77.
[6] T. Yoshinari, R.T. Forbes, P. York, Y. Kawashima, The improved compaction properties of mannitol after a moisture-induced polymorphic transition, International Journal of Pharmaceutics, 258 (2003) 121-131.
[9] H. Leonhard Ohrem, E Schornick, A Kalivoda & R Ognibene (2014) Why is mannitol becoming more and more popular as a pharmaceutical excipient in solid dosage forms?, Pharmaceutical Development and Technology, 19:3, 257-262, DOI: 10.3109/10837450.2013.775154.
In a pioneering procedure, a team of surgeons at New York University Langone Health Grossman School of Medicine in New...

[bsa_pro_ad_space id=2]
[bsa_pro_ad_space id=4]
PharmaCentral.com may on occasion publish user-generated content. Any information provided on our platform is for general informational and educational purposes only. All information is provided in good faith to enable collaboration and sharing of know-how among our community of users. Authors who submit content retain copyright to it.
PharmaCentral.com does not make any representation or warranty of any kind regarding its accuracy, adequacy, or legality. Any references to particular product names, brands, descriptions, formats, styles, corporate entities, tests, applications, technologies, uses, standardisations, medical conditions, and treatments are for illustration purposes and should not be considered complete or binding. All respective intellectual property, such as trademarks and logos, are properties of their owners
Under Section 107 of the Copyright Act 1976, allowance is made for ‘Fair Use’ for purposes such as criticism, comment, news, reporting, scholarship, education, and research.
Fair use is a use permitted by copyright statute that might otherwise be infringing.
Some information contained on PharmaCentral.com may contain copyrighted material, the use of which may not have been specifically authorised by the respective copyright owners. Some material is made available to help explain and relay complex phenomena, formulae, physical and chemical constants, and other concepts that are scientifically incontestable but relevant to the use of products, and/or to illustrate, transmit, and teach pharmaceutical science principles. Some material is published to support research and user education, and for the public good.