Opinion | What Does the Recent Pig-to-Human Kidney Transplant Mean for Tissue Therapeutics?
In a pioneering procedure, a team of surgeons at New York University Langone Health Grossman School of Medicine in New...
Hydrophobic Colloidal Silicon Dioxide is a surface-modified synthetic amorphous silica that is differentiated from standard synthetic amorphous silica (e.g Colloidal silicon dioxide) in having its surface-based silanol groups bonded to dimethyl silyl groups making it hydrophobic in character. Hydrophobic colloidal silica occurs as a light, fine, white or almost white amorphous powder, not wettable by water.
Synonyms and Trade Names: Hydrophobic Colloidal Anhydrous Silica; Silica, Hydrophobic Colloidal; Hydrophobic Colloidal Silica; Hydrophobic Fumed Silica; Silica Dimethyl Silylate; Hydrophobic Fumed Silica; Silica Dimethyl Silylate; Silicic Acid, Silanated; Silicon Dioxide, Silanated; AEROSIL® R972; HDK® H15
Uses and Applications: Anticaking Agent; Emulsion Stabilizer; Glidant; Suspending Agent; Thermal Stabilizer, and Viscosity-increasing Agent
Hydrophobic colloidal silicon dioxide is a synthetic amorphous silica derivative in which the surface of the fumed silica particle has been modified by the addition of dimethyl silyl groups. The surface modification is achieved via a controlled chemical process that involves the attachment of dimethyl silyl groups, rendering the silica less wettable. It is approved for use in pharmaceutical products as an excipient and is supplied as a light, fine, white or almost white amorphous fluffy powder.
Hydrophobic colloidal silica occurs as a light, fine, white or almost white amorphous powder, not wettable by water.
Silicon and oxygen are the two most abundant elements in the earth’s crust. In nature, silicon almost always exists in combination with oxygen, either as free silica (SiO2), in conjunction with other elements (for instance, in silicates, which are the main minerals in rocks and soil), or as combined silica (SiO3). The different silicon compounds have substantially different chemical properties, applications, and hazards.
Free silica (Silicon dioxide) is a hard, low-reactivity, colourless substance that occurs naturally in rocks and minerals or can also be industrially produced in the form of synthetic amorphous silica. All forms of Silica, whether natural, synthetic, crystalline, cryptocrystalline or amorphous, are assigned a single CAS Registry number [7631-86-9]. For convenience, the different forms of Silica can be divided into three main groups as shown in the chart below:
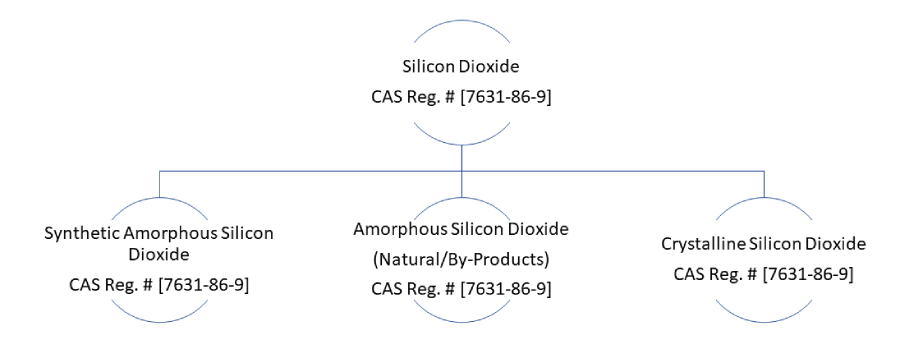
At the fundamental level of Silica’s chemical properties is the Silica tetrahedron (SiO4), which consists of a central Silicon cation covalently bonded to four oxygen atoms, arranged in the shape of a tetrahedron. Silica tetrahedra may be linked and arranged in a variety of ways, from simple to complex three-dimensional frameworks. Crystalline forms of Silica exhibit a highly ordered crystal lattice, determined by the ordered arrangement of the Silica tetrahedra. Amorphous forms, on the other hand, have random, disordered lattices. The orientation of the bonds is random, and there is no long-range periodicity.
To differentiate between the different silica analogues, new CAS Registry numbers have been assigned. These are shown in the chart below:
![]()
Synthetic amorphous silicon dioxides are further divided into three main types, namely:
Even though they share the same chemical structure and synthetic origin, Synthetic silicas exhibit different properties, as briefly reviewed below:
Pyrogenic silicon dioxide is produced using a high-temperature process in which silicon tetrachloride is vaporised in an oxygen-hydrogen atmosphere according to the following chemical reaction:
SiCl4 + 2H2 + O2 -> SiO2 + 4HCl
The raw materials used in the silica production process are all inorganic and very pure. As a result, the synthesis produces only hydrochloric acid (which is easily removed) and silica in very high states of purity (typically > 99.9%). Silicon dioxide produced pyrogenically exists in the form of chain-like, branched aggregates, giving rise to a fluffy, light powder. (The term “fume” alludes to the method of manufacture, which involves the use of a flame.) Varying processing conditions allows the production of silica products with different specific surface areas, typically between 50 and 400 m2/g.
The pyrogenic method for producing Silica was invented in 1941 by Harry Klopfer, a scientist at Degussa (now part of Evonik AG). This method is what is still used by Evonik (for the production of pyrogenic silica marketed under the AEROSIL® brand name) and Cabot Corporation (for the Cab-O-sil® fumed silica brand). Note that these silica grades can be used in their native (unprocessed) state. They can also be further processed (for example, spray drying, granulation, or surface chemical modification) to turn them into other technical silica grades.
Precipitated silicon dioxide is silica produced in an aqueous solution at temperatures >60 °C. In this process, sodium silicate (waterglass) undergoes controlled neutralisation with either concentrated sulphuric acid or hydrochloric acid. The Silica precipitates out as a slurry of (hydrated) silica, which is washed and filtered to remove by-products. It is then dried in hot air and milled or passed through a classifier.
Precipitated silicon dioxide has been known since the mid 17th century. It was not until the 1920s that its practical uses and industrial production were fully established. Currently, Precipitated silica is produced in volumes that are up to x10 greater than for Pyrogen silica. The method and conditions have been fine-tuned and now permit the production of many types of synthetic silica that are structurally and characteristically different, including exhibiting internal pore volume/specific surface area, larger particle sizes, and water content.
3). Surface-Modified Silicon Dioxide
The Silica grades described thus far are available for use in their native or unmodified state. These materials have freely accessible silanol groups (Si-OH) on the surfaces of Silica particles, rendering them hydrophilic. Frequently, it is desirable to have hydrophobic silica, i.e a product that repels water. Hydrophobicity can be achieved through a post-synthesis step in which the silanol groups are reacted with organic groups. The added organic groups are tightly bound to the surface (via covalent bonds) and are only broken via thermal decomposition.

Pharmaceutical-approved hydrophobic silica is produced by reacting hydrophilic silicon dioxide with dimethylchlorosilane immediately after the production of Silica particles in the hydrogen flame chamber. This process is also conducted at high temperatures and allows dimethysilyl groups to be bound irreversibly onto the surface of the silica via siloxane bonds. This produces a material that, while appearing identical to the precursor Silica, is very hydrophobic, repels water and does not absorb moisture from the environment.
“Colloidal” is used in reference to both pyrogenic and precipitated silica. It may be confused with Silica colloids, which are also obtained via the wet chemical route. Note that the International Union of Pure and Applied Chemistry (IUPAC) defines colloids as systems (dispersions) in which particles of colloidal size (1 nm–1000 nm) of any nature (solid, liquid, or gas) are dispersed in a continuous phase of different composition or state.
Thus, in the strictest sense, the term ‘colloidal silica’ applies to stable dispersions (or sols) consisting of discrete particles of amorphous silica having sizes of between 5 and 100 nm. These colloidal silicas are commercially available in the form of sols or dried powders (e.g., xerogels, dry precipitates, aerogels, or calcinated coarcervates). In a broad sense, however, many other forms of silica (other than wet or dry silica sols above) are colloidal on the grounds that they are composed of particles in a colloidal state of subdivision (1-1000 nm). Moreover, the silica particles and aggregates are self-supporting and stabilised dispersions of silica particles in a continuous air phase and are unaffected by gravitational forces. Finally, fumed silica is commonly referred to as ‘colloidal’ because the silica powders are made by condensing a silica precursor from a vapour phase. In this sense, fumed silica particles are dispersed in a gas during its production process.
The SiO2 molecules in synthetic silicon dioxide do not exist in isolation. While the primary structure is the tetrahedron, consisting of one silicon atom bonded to four oxygen atoms, tetrahedrons arrange into networks. During the synthesis process, minute droplets of SiO2 initially aggregate into so-called nuclides, which combine stochastically into even larger particles, facilitated by weak physical interactions such as van der Waal’s forces.
Another important property of synthetic silica is its specific surface area. Fumed silicon dioxide, in particular, has only one surface, which is external and little or non-existent internal pore volume. Precipitated silica, on the other hand, is mesoporous and exhibits an internal surface. Generally, the higher the specific surface area, the greater the degree of agglomeration. It is these aggregates that partly contribute to the unique functionalities of amorphous silicon dioxide.

| Chemical Name | Silica dimethyl silylate |
| CAS Registry Number | [68611-44-9] |
| Empirical Formula | Si02 (partly alkylated for hydrophobation) |
| Molecular Weight | 60.08 |
| EC/EINECS Number | 271-893-4 |
| UNII Code (FDA) | EU2PSP0G0W |
Hydrophobic silicon dioxide is approved for use as a pharmaceutical excipient and can be used in pharmaceuticals in Europe and in the USA. It is also approved by FDA and Europe for food contact articles and in feed additives (E551b). It is not currently approved for use as an excipient in Japan.
| Physical form | Solid, powder |
| Appearance | White, fluffy powder |
| Flowability | Poorly flowing and compressible |
| Bulk density | 0.094 g/ml (AEROSIL® R972 Pharma – Evonik) |
| Tapped density | 0.115 g/ml (AEROSIL® R972 Pharma – Evonik) |
| Porosity | Not specified |
| Moisture content | <0.5% w/w |
| Particle size distribution | The primary particle size is 16 nm for AEROSIL® R972 Pharma. Forms loose agglomerates of 10 -200µm |
| Solubility | Repels water. Solubility in water, organic solvents and mineral acids is negligible |
| Specific gravity | 2.0 – 2.2 |
| Specific surface area | 90 – 110 m2/g |
| USP-NF | Ph.Eur | B.P | |
| Name | Hydrophobic Colloidal Silica | Silica, Hydrophobic Colloidal | Hydrophobic Colloidal Anhydrous Silica |
| Authorised Uses | Excipient | Excipient | Excipient |
| Definition | specified | specified | specified |
| Characters | specified | specified | specified |
| Identification | specified | specified | specified |
| Solubility | ≤0.5% | ≤1.0% | specified |
| Chlorides | ≤0.025% | ≤250 ppm | ≤250 ppm |
| Water-dispersible fraction | ≤ 3.0 % | ≤ 3.0 % | ≤ 3.0 % |
| Loss on Ignition (%) | ≤ 6.0% | ≤ 6.0% | ≤ 6.0% |
| Heavy Metals (Lead) | 0.0025% | ≤ 25 ppm | ≤ 25 ppm |
| Assay (%) | 99.0-101.0 | 99.0-101.0 | 99.0-101.0 |
| Labelling | specified | n/a |
Key: n/a Specification is Not listed
*All claims in respect to the conformity are subject to our Terms and Conditions. No express or implied warranty is made for specific properties or fitness for any particular application or purpose.
Hydrophobic colloidal silica is used as an aticaking agent; emulsion stabilizer; glidant; suspending agent; thermal stabilizer and viscosity-increasing agent. Hydrophobic colloidal silica has nano-sized primary particles and a large specific surface area, which provide desirable flow characteristics in powder blends such as those used in tableting and capsule filling, especially those liable to adsorb surface moisture. Since hydrophobic grades absorb less moisture they may be of particular interest for formulations that are sensitive to moisture.
Another important use of Hydrophobic colloidal silica is in the control of the viscosity of liquid formulations (via thixotropy), which can be utilised for thickening and stabilising emulsions, or as a suspending agent in gels and some types of semisolid preparations. Although Hydrophobic colloidal silica is less effective for altering the viscosity of aqueous media, it can still be used to increase the viscosity of and stabilize the oily phase of a w/o emulsion. When added to formulation ingredients that have a similar refractive index, transparent gels can be obtained.
Hydrophobic silica is approved for use in pharmaceuticals in Europe and in the USA. It is also approved by FDA and Europe for food contact articles and in feed additives (E551b). Hydrophobic silica is not approved for use in food intended for human consumption. It is permitted in food contact articles in Europe (listed as silicon dioxide, silanated) and as an anticaking agent in animal feed mineral premixes in Europe and the USA. The safety profile of hydrophobic colloidal silica is the same as for the hydrophilic silica types, as the modified silica surface does not change toxicological profile of the material.
Toxicology: LD50 (rat, IV): 0.015g/kg. Carcinogenicity: Not established
Hydrophobic colloidal silica should be stored in a well-closed container. It will not absorb moisture but may still absorb volatile substances owing to its high surface area. It has a shelf life of 12 months.
When handling the product, you should observe institutional SHEQ protocols appropriate to the circumstances and quantity of the material. Eye protection and gloves are recommended. Inhalation of Hydrophobic colloidal silica dust may cause irritation of the respiratory tract (although it not not associated with fibrosis of the lungs). If suitable containment facilities are absent in the facility, a dust mask should be used when dealing with small quantities and for larger volumes, a dust respirator is recommended.
A sustainability score for Hydrophobic silicon dioxide has not yet been provided.
[1] Evonik Industries, AEROSIL(R) Pharma Colloidal silicon dioxide, Technical information.
[2] Evonik Industries, SIPERNAT and AEROSIL – an essential in industrial powder technology, Technical information.
[3] World Health Organization & Food and Agriculture Organization of the United Nations. (1991). Evaluation of certain food additives and contaminants : thirty-seventh report of the Joint FAO/WHO Expert Committee on Food Additives [meeting held in Geneva from 5 to 14 June 1990], (1991).
[5] G.K. Bolhuis, N. Anthony Armstrong, Excipients for direct compaction—an update, Pharmaceutical Development and Technology, 11 (2006) 111-124.
[6] L.C. Becker, W.F. Bergfeld, D.V. Belsito, R.A. Hill, C.D. Klaassen, D. Liebler, J.G. Marks, R.C. Shank, T.J. Slaga, P.W. Snyder, F.A. Andersen, Safety Assessment of Silylates and Surface-Modified Siloxysilicates, international Journal of Toxicology, 32 (2013).
[7] D. Majerová, L. Kulaviak, M. Růžička, F. Štěpánek, P. Zámostný, Effect of colloidal silica on rheological properties of common pharmaceutical excipients, European Journal of Pharmaceutics and Biopharmaceutics, 106 (2016) 2-8.
[8] Sccs, P.H.M. Hoet, Opinion of the Scientific Committee on Consumer Safety (SCCS) – Revision of the opinion on the safety of the use of Silica, Hydrated Silica, and Silica Surface Modified with Alkyl Silylates (nano form) in cosmetic products, regulatory toxicology and pharmacology, 74 (2016) 79-80.
[9] T.U.S.P. Convention, Food Chemicals Codex (11th Edition), (2018).
In a pioneering procedure, a team of surgeons at New York University Langone Health Grossman School of Medicine in New...

[bsa_pro_ad_space id=2]
[bsa_pro_ad_space id=4]
PharmaCentral.com may on occasion publish user-generated content. Any information provided on our platform is for general informational and educational purposes only. All information is provided in good faith to enable collaboration and sharing of know-how among our community of users. Authors who submit content retain copyright to it.
PharmaCentral.com does not make any representation or warranty of any kind regarding its accuracy, adequacy, or legality. Any references to particular product names, brands, descriptions, formats, styles, corporate entities, tests, applications, technologies, uses, standardisations, medical conditions, and treatments are for illustration purposes and should not be considered complete or binding. All respective intellectual property, such as trademarks and logos, are properties of their owners
Under Section 107 of the Copyright Act 1976, allowance is made for ‘Fair Use’ for purposes such as criticism, comment, news, reporting, scholarship, education, and research.
Fair use is a use permitted by copyright statute that might otherwise be infringing.
Some information contained on PharmaCentral.com may contain copyrighted material, the use of which may not have been specifically authorised by the respective copyright owners. Some material is made available to help explain and relay complex phenomena, formulae, physical and chemical constants, and other concepts that are scientifically incontestable but relevant to the use of products, and/or to illustrate, transmit, and teach pharmaceutical science principles. Some material is published to support research and user education, and for the public good.