Opinion | What Does the Recent Pig-to-Human Kidney Transplant Mean for Tissue Therapeutics?
In a pioneering procedure, a team of surgeons at New York University Langone Health Grossman School of Medicine in New...
Polyoxyethylene Castor Oil Derivatives are synthetic non-ionic surfactants obtained by reacting varying amount of ethylene oxide and either castor oil or hydrogenated castor oil. Polyoxyethylene Castor Oil derivatives are designated by a number (n= 30-60) representing the number of oxyethylene units in the compounds. They are supplied either as pale-yellow oily liquids or white to yellowish, semisolid pastes.
Pharmacopoeial Compliance: USP-NF; Ph.Eur; I.P; B.P
Synonyms and Trade Names: Polyoxyethylene Castor Oil; Polyoxyl Castor Oil; Macrogoiglycerol Ricinoleate; Castor Oil Ethylene Glycol Polymer; Glycerol Polyethylene Glycol Ricinoleate; Ethoxylated Castor Oil; Polyoxyl 35 Castor Oil; Arlatone®; Cremophor® EL35; Eumulgin®, Lipocol®; Kolliphor® EL 35; Simulsol®
Uses and Applications: Emulsifying Agent; Solubilising Agent and Wetting Agent
Polyoxyethylene Castor oil is a generic name for various closely-related PEGylated Castor oil derivatives. They are obtained by reacting varying amounts of ethylene oxide with Castor oil. Individual substances are assigned a numerical suffix (X), representing the number of stoichiometric equivalents of ethylene oxide added to one stoichiometric equivalent of Castor oil.
As non-ionic surfactants, Polyoxyethylene Castor oils are widely used in pharmaceuticals formulations, fragrance products, hair sprays, deodorants, and indoor tanning preparations. The hydrophobic portion of the molecule is represented by the fatty acid esters of glycerol (Glycerol Polyethylene glycol oxystearate, together with fatty acid glycerol polyglycol esters) while the Polyethylene glycol portion represents the hydrophilic portion.
Just like natural glycerides, Polyoxyethylene Castor oil derivatives occur as complex mixtures made up of closely-related components, primarily Polyethylene glycol conjugates, as well as fatty acid esters of Polyethene glycol and ethoxylated glycerol. Ethoxylation of glycerides occurs not only as an etherification of the free alcohol groups of the glycerides but also as a transesterification, which leads to a net insertion of Polyethylene glycol groups between the Glyceryl and fatty acid components of the glyceride. In addition, a proportion of the ethylene oxide also reacts with other molecules of ethylene oxide, resulting in some Polyethylene glycols remaining unattached to glycerides or acid groups. This is why Hydrogenated Castor oil triglycerides are complex mixtures of structurally related molecules.
In theory, X, the numerical suffix assigned to Polyoxyethylene Castor oil derivates can vary between 2 and 200, i.e Polyxyl 2, 3, 4, …. 200 Castor oil. In reality, there are only around nine different derivatives available commercially, as shown in the table below.
| Polyoxyl-9 Castor Oil | Polyoxyl 9 Castor Oil is a Polyethylene glycol derivative of Castor Oil with an average of 9 moles of ethylene oxide. It consists of a mixture of the etherification and esterification products of Castor oil glycerides and fatty acids from castor oil, with nine equivalents of ethylene oxide |
| Polyoxyl-25 Castor Oil | Polyoxyl 25 Castor Oil is a Polyethylene glycol derivative of Castor Oil with an average of 25 moles of ethylene oxide. It consists of a mixture of the etherification and esterification products of castor oil glycerides and fatty acids from Castor oil, with twenty-five equivalents of ethylene oxide |
| Polyoxyl-30 Castor Oil | Polyoxyl 30 Castor Oil is a Polyethylene glycol derivative of Castor Oil with an average of 30 moles of ethylene oxide. It consists of a mixture of the etherification product of castor oil glycerides and the esterification product of the fatty acids from Castor oil, with one end of a Polyethylene glycol chain, averaging thirty ethylene glycol repeat units in length |
| Polyoxyl-33 Castor Oil | Polyoxyl 33 Castor Oil is a Polyethylene glycol derivative of Castor Oil with an average of 33 moles of ethylene oxide. It consists of a mixture of the etherification product of castor oil glycerides and the esterification product of the fatty acids from Castor oil, with one end of a Polyethylene glycol chain, averaging thirty- three ethylene glycol repeat units in length |
| Polyoxyl-35 Castor Oil | Polyoxyl 35 Castor Oil is a Polyethylene glycol derivative of Castor Oil with an average of 35 moles of ethylene oxide. It consists of a mixture of the etherification and esterification products of castor oil glycerides and fatty acids from castor oil, with thirty-five equivalents of ethylene oxide. |
| Polyoxyl-36 Castor Oil | Polyoxyl 36 Castor Oil is a Polyethylene glycol derivative of Castor Oil with an average of 36 moles of ethylene oxide. It consists of a mixture of the etherification and esterification products of castor oil glycerides and fatty acids from Castor oil, with thirty-six equivalents of ethylene oxide. |
| Polyoxyl-40 Castor Oil | Polyoxyl 40 Castor Oil is a Polyethylene glycol derivative of Castor Oil with an average of 40 moles of ethylene oxide. It consists of a mixture of the etherification and esterification products of castor oil glycerides and fatty acids from Castor oil, with forty equivalents of ethylene oxide. |
| Polyoxyl-50 Castor Oil | Polyoxyl 44 Castor Oil is a Polyethylene glycol derivative of Castor Oil with an average of 44 moles of ethylene oxide. It consists of a mixture of the etherification and esterification products of castor oil glycerides and fatty acids from Castor oil, with forty-four equivalents of ethylene oxide. |
| Polyoxyl-60 Castor Oil | Polyoxyl 60 Castor Oil is a Polyethylene glycol derivative of Castor Oil with an average of 60 moles of ethylene oxide. It consists of a mixture of the etherification and esterification products of castor oil glycerides and fatty acids from Castor oil, with sixty equivalents of ethylene oxide. |
In the Ph.Eur, only derivatives ethoxylated with 30-50 moles of ethylene oxide are officially recognised. In contrast, the USP-NF lists a single monograph for Polyoxyl 35 Castor oil. Both compendia recognise the complex nature of these substances, describing them as containing mainly the triricinoleate esters of ethoxylated glycerol with small amounts of Polyethylene glycol ricinoleate and free glycols. Polyoxyethylene Castor oil derivatves are supplied as either as pale-yellow oily liquids or white to yellowish, semisolid pastes.
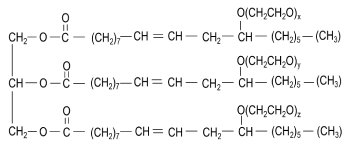
x + y + z = ≈ n
| Chemical Name | Castor oil, ethoxylated |
| CAS Registry Number | [161791 12-6] |
| Empirical Formula | (C2H4O)0-3(C2H4O)0-3(C2H4O)0-3(C2H4O)0-3(C2H4O)0-3(C2H4O)0-3C57H104O9 |
| Molecular Weight | 1113.5861 |
| EC Number | 500-151-7 |
| UNII Code (FDA) | 6D4M1DAL60 |
Several Polyoxyethylene Castor oil derivatives are available commercially, however not all of them are officially listed in the pharmacopoeia, and therefore, recognised as excipients. In the Ph.Eur monograph, Polyoxyl Castor oil is restricted to grades that contain ricinoleyl glycerol ethoxylated with 30–50 molecules of ethylene oxide, i.e Polyoxyl 30 Castor oil, Polyoxyl 33 Castor oil, Polyoxyl 35 Castor oil, Polyoxyl 36 Castor oil, Polyoxyl 40 Castor oil and Polyoxyl 50 Castor oil. The USP-NF contains a single monograph which corresponds to Polyoxyl 35 Castor oil. Polyoxyl 35 Castor oil is included in the FDA Inactive Ingredients Database (covering IV injections and ophthalmic solutions).
Polyoxyethylene castor oil derivatives are complex mixtures of various hydrophobic and hydrophilic components. Depending on degrees of ethoxylation (moles)/PEG units as indicated by their numerical suffix (n), the physicochemical properties of Polyoxyethylene Castor oil derivatives vary considerably. Below is a summary of the key physicochemical properties for Polyoxyl 35 Castor oil:
| Form | Pale-yellow oily liquids or white to yellowish, semisolid pastes |
| Appearance | Pale yellow oily liquid, clear above 26 oC |
| Acid value | ≤2.0 |
| HLB value | 12 – 14 |
| Hydroxyl value | 65 – 78 |
| Iodine value | 25 – 35 |
| Saponification value | 65 – 70 |
| Water content % | 2.8 |
| Melting point (oC) | 19- – 20 |
| Solidification point (oC) | – |
| Density | 1.05-1.06 g/ml |
| pH | 6-8 |
| Refractive Index | 1.471 |
| Surface tension | 40.9 mN/m |
| Viscosity (25 oC) | 600-800 cSt |
| Solubility | Soluble in Water, Alcohol (95%), Castor oil, Fatty Acids, and Olive oil. Insoluble in Mineral oil |
| Test | USP-NF | Ph.Eur |
| Official name | Polyoxyl 35 Castor Oil | Macrogolglycerol Ricinoleate |
| Authorised used | Excipient | Excipient |
| Definition | specified | specified |
| Identification | A
B C |
A
B C D |
| Characters | n/a | specified |
| Appearance of solution | n/a | specified |
| Alkalinity | n/a | n/a |
| Specific gravity | 1.05-1.06 | n/a |
| Viscosity at 25 oC | 600-850 mPa s | n/a |
| Water | ≤3.0% | ≤3.0% |
| Residue on ignition/Total ash | ≤0.3% | ≤0.3% |
| Heavy metals | ≤10ppm | ≤10ppm |
| Acid value | ≤2.0 | ≤2.0 |
| Hydroxyl value | ≤65-80 | ≤65-80 |
| Saponification value | ≤60-75 | ≤60-75 |
| Ethylene glycol | ≤620 µg/g | ≤1 pmm |
| Diethylene glycol and Ethylene glycol | ≤2500 µg/g | n/a |
| 1,4-Dioxan | ≤10 | n/a |
| Free ethylene oxide | ≤1 µg/g | ≤1ppm |
| Assay | n/a | n/a |
| Labelling | n/a | specified |
Polyoxyethylene Castor oil derivatives are synthetic non-ionic surfactants used in liquid oral, topical, and parenteral pharmaceutical formulations where they function as emulsifying agents, solubilizing agents and wetting agents. In cosmetic formulations, Polyoxyl 30, -33, -35, -36, and -40 Castor oil function primarily as surfactants and can be used in concentrations of up to 50%.
In pharmaceutical products, Polyoxyl 35 Castor oil is the derivative most commonly encountered. It has been used to solubilise lipidic and/or poorly soluble drugs, including emulsification of fat-soluble vitamins, A, D, E, and K. Polyoxyl 35 Castor oil has been used to successfully formulate a wide variety of BCS IV hydrophobic drugs, including anaesthetics, photosensitisers, sedatives, immunosuppressive agents and (experimental) anticancer drugs which had previously eluded conventional formulation efforts.
Following the discovery of the anticancer agent, Paclitaxel, the development of the drug product was severely hampered for more than a decade due to its poor solubility in most conventional solvents. A viable formulation consisting of 50% Polyoxyl 35 Castor oil and 50% dehydrated Ethanol has successfully been commercialised as TAXOL® (Paclitaxel) – Bristol-Myers Squibb Company.
In veterinary pharmaceuticals, Polyoxyl 35 Castor oil is also utilised to emulsify fish oils and oils fats that are incorporated into animal feeds. In this sense, it aids the bioavailability of nutritive and therapeutic substances, thereby improving their efficacy.
In personal care products, Polyoxyl 35 castor oil is mainly used as a solubilizing agent for flavouring (e.g mouthwashes), perfumes and volatile oils. As a general rule, when Polyoxyl 35 Castor oil is added in concentrations above 10%, clear or opalescent liquids are obtained. At lower concentrations, however (5-10%), emulsions can form when water is added to the system.
Polyoxyethylene Castor oil derivatives are widely used in a variety of oral, topical, and parenteral pharmaceutical products as well as personal care formulations. They have been studied to determine their safety in a number of acute and chronic toxicity tests in animal models. These studies have shown Polyoxyethylene Castor oil derivatives to be essentially non-toxic and non-irritant excipients. Data on toxicokinetics, single-dose and repeated-dose toxicity, reproductive and developmental toxicity, genotoxicity, carcinogenicity, dermal and ocular irritation, and sensitization are available for Polyoxyethylene Castor oils. Reports of anaphylactic reactions, cardiotoxicity, nephrotoxicity, neurotoxicity and pulmonary toxicity have also been observed in humans and animals following parenteral administration of formulations containing these substances.
Polyoxyl Castor oil derivatives are stable and non-reactive substances. The assigned shelf-life is 2 years if the material is stored in the original container at 25 oC. Aqueous solutions of Polyoxyl Castor oil are stable even when exposed to moderate heat stress. Prolonged exposure can cause phase separation although the product can be restored to its original form by homogenization. In an aqueous solution, Polyoxyl Castor oil derivatives remain stable even when electrolytes are present in the solution. A number of organic substances may cause precipitation at certain concentrations, especially compounds containing phenolic hydroxyl groups.
Polyoxyethylene castor oil derivatives should be stored in a well-closed, airtight container, protected from light, in a cool, dry place. When handling Polyoxyl Castor oil derivatives, workers should observe applicable SHEQ protocols appropriate to the circumstances and quantity of material handled. the use of eye protection and gloves are recommended.
A sustainability score for Polyoxylated Castor oil derivates has not been availed.
In a pioneering procedure, a team of surgeons at New York University Langone Health Grossman School of Medicine in New...

[bsa_pro_ad_space id=2]
[bsa_pro_ad_space id=4]
PharmaCentral.com may on occasion publish user-generated content. Any information provided on our platform is for general informational and educational purposes only. All information is provided in good faith to enable collaboration and sharing of know-how among our community of users. Authors who submit content retain copyright to it.
PharmaCentral.com does not make any representation or warranty of any kind regarding its accuracy, adequacy, or legality. Any references to particular product names, brands, descriptions, formats, styles, corporate entities, tests, applications, technologies, uses, standardisations, medical conditions, and treatments are for illustration purposes and should not be considered complete or binding. All respective intellectual property, such as trademarks and logos, are properties of their owners
Under Section 107 of the Copyright Act 1976, allowance is made for ‘Fair Use’ for purposes such as criticism, comment, news, reporting, scholarship, education, and research.
Fair use is a use permitted by copyright statute that might otherwise be infringing.
Some information contained on PharmaCentral.com may contain copyrighted material, the use of which may not have been specifically authorised by the respective copyright owners. Some material is made available to help explain and relay complex phenomena, formulae, physical and chemical constants, and other concepts that are scientifically incontestable but relevant to the use of products, and/or to illustrate, transmit, and teach pharmaceutical science principles. Some material is published to support research and user education, and for the public good.