| Weight | 5 kg |
|---|
Strawberry Flavour Excipient | Uses, Suppliers, and Specifications
Strawberry flavour denotes blends of approved and well-defined chemical substances and extracts with a sweet, fruity and strawberry olfactory and gustatory profile for use in oral pharmaceutical products as flavourings.
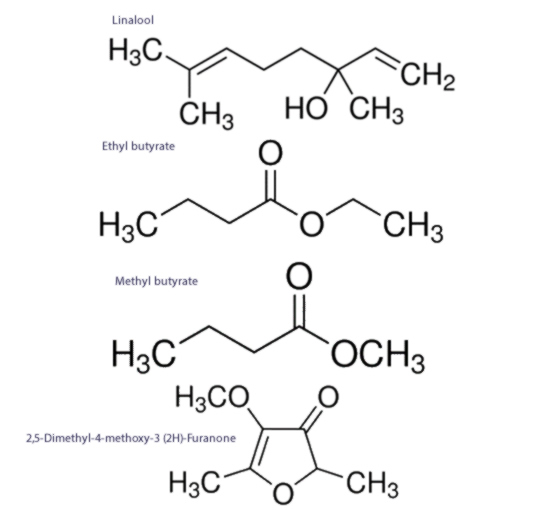
Strawberry plants exhibit considerable chemical diversity, with up to 81 different compounds cited as being pertinent to strawberry flavour. While there is no consensus on which substances are flavour defining, a select few substances are considered to positively correlate with strawberry flavour intensity, and include linalool; ethyl butyrate; methyl butyrate; and 32,5-Dimethyl-4-methoxy-3 (2H)-furanone,
Note that when ‘Natural Strawberry flavouring’ is specified, this usually denotes flavouring substances naturally identified in nature and obtained by appropriate physical, enzymatic or microbiological steps from material of plant origin. On the other hand, ‘Artificial Strawberry flavouring’ denotes any flavouring substances that are not defined as natural, even if they have the exact same chemical composition as those isolated directly from nature. The distinction between natural or artificial flavourings has no bearing on how safe, healthy, or appealing they are.
Physicochemical Properties
Powdered Strawberry Flavour
| Appearance | Solid, powder |
| Colour | Cream, white, characteristic |
| Odour | Characteristic |
| Taste | Characteristic |
| Extractable oil | ≤ 10% |
| Particle size 0.85mm | ≤ 99.0% passes |
| Water content | 0.0 – 10.0% |
| Flash Point | >100 oC |
| Aerobic Plate Count (CFU) | Max 1000/g |
| Coliforms/Enterobacteriacea (CFU) | Max 10/g |
| Mould (CFU) | Max 100/g |
| Yeast (CFU) | Max 100/g |
| Salmonella | Absent |
| E.Coli | Absent |
Liquid Strawberry Flavour
| Appearance | Liquid, Limpid Liquid |
| Colour | Colourless to pale yellow |
| Odour | Characteristic |
| Taste | Characteristc |
| Extractable oil | Varies |
| Water content | 0.0 – 10.0% |
| Flash Point | <150 oC |
| Specific gravity | 1.00- 1.100 |
| Refractive Index | 1.40- 1.60 |
| Aerobic Plate Count (CFU) | Max 1000/g |
| Coliforms/Enterobacteriacea (CFU) | Max 10/g |
| Mould (CFU) | Max 100/g |
| Yeast (CFU) | Max 100/g |
| Salmonella | Absente |
| E.Coli | Absent |
Applications in Pharmaceutical Formulations
Strawberry flavourings are available in liquid or powder form, in a variety of concentrations, formulations and formats (spray-dried or flexarome).
Strawberry flavourings are added to pharmaceutical products for various reasons, but mainly
- to impart flavour to an otherwise bland product
- to impose a different flavour character from that arising from the basic ingredients
- to boost weak intrinsic flavours or replace flavour notes lost during processing
- to modify or complement an existing flavour profile
- to disguise or cover undesirable flavour attributes
- to overcome seasonal variability ‘in natural flavouring materials or constituents
- to impart a flavour where the use of a natural flavouring material is technologically impracticable
One of the major functions of intentionally added flavourings is to extend a product’s scope and range, but their specific application is determined by factors which are not exclusively technical. These include:
- Acceptability to the consumer – often the flavour is open to wide hedonic interpretation, with preferences for a particular flavouring dependent on such factors as ethnicity, education, age, environment, and even personal moods. The strength and quality of flavours are also often regionalised. This can be challenging to product manufacturers aiming at international market.
- Regulatory acceptability – the use of flavourings has become highly regulated order to safeguard consumers from real or supposed health hazards arising from the ingestion of materials intentionally added to the natural diet. Regulators also want to prevent deception as to the true nature of the products which the consumer must take on trust. It is essential that any product complies with the legislation of the country in which it is offered for sale.
- The nature of the product – a major result in advances in processing technology, imaginative product conception, versatile packaging and efficient distribution is wider range of consumable products and the formats in which flavourings can be incorporated. Suffice to say, dry products call for powdered flavourings and wet products enable one to use flavourings in liquid form.
Pharmacopoeial Specifications
Compliance of flavouring constituents with any specific pharmacopoeia is not required.
Safety and Regulatory Status
From a regulatory point of view, there are no specific regulations or positive/negative lists on flavourings for pharmaceutical products and by default, when checking flavourings acceptability of flavours, reference is made to food regulations of the relevant country.
It is important to bear in mind that whether flavoured or non-flavoured, registration procedures for pharmaceutical products, both OTC and prescribed, apply, as laid down in Directive 2001/83/EC.
Although both qualitative and quantitative composition of the medicinal product is required, for flavours, it is only necessary to mention the main constituents, with an appropriate method of identification, such as gas chromatograph or infrared spectroscopy, as appropriate.
For paediatric formulations, the following restrictions apply:
| Excipient | Role in formulation | Potential Adverse effect(s) |
| Propylene glycol | Solvent | Central nervous system (CNS) effects, especially in neonates and children under four years |
| Ethanol | Solvent | Intoxication |
| Polyoxyl castor oil | Vehicle | Severe anaphylactic reactions |
| Polysorbate 80 | Solubilising agent | E-Ferol syndrome, hypersensitivity reactions |
| Benzyl alcohol | Preservative | ‘Gasping syndrome’ in neonates |
| Benzoic acid | Preservative | Jaundice in neonates |
| Parabens (methyl-, ethyl- and propyl-hydroxybenzoates) | Preservative
|
Suggestion of oestrogenic activity with potential reproductive effects (with propylparaben), hypersensitivity reactions, hyperbilirubinaemia in neonates |
| Benzalkonium chloride | Preservative | Bronchospasm from anti-asthmatic drugs |
| Sodium metasulfites | Antioxidant | Wheezing, dyspnoea and chest tightness in asthmatic children |
| Sorbitol | Sweetener | Osmotic diarrhoea and gastrointestinal discomfort |
| Mannitol | Sweetener | Osmotic diarrhoea and gastrointestinal discomfort |
| Glucose and sucrose | Sweetener | Obesity and tooth decay |
| Saccharin | Artificial sweetener | Hypersensitivity and photosensitivity reactions |
| Aspartame | Artificial sweetener | A source of phenylalanine that should be avoided in patients with phenylketonuria |
| Colourants | Colouring agents | Sensitivity reactions and hyperactive behaviour in children[ |
Note that flavourings are considered as ‘single’ excipients from a regulatory point of view even though they consist of several ingredients. Indeed, most flavourings contain multiple components which in their own right are considered as excipients and therefore require labelling.
Stability and Storage Conditions
Liquid flavours are volatile and preparations may exhibit thermodynamic instability. The prolonged action of heat may induce physical separation into separate phases. Most liquid flavours are stable for at least 12 months if stored in unopened airtight containers at room temperature (<25 0C).
The pH may drop slightly during storage, which should be taken into account. Separation into phases may also occur, but agitating the hot solution can reverse this.
Flavour powder should be stored in an airtight, corrosion resistant container and protected from moisture. The use of glass, plastic, or resin-lined containers is recommended for the storage of formulations containing carbomer.
Handling Precautions
Observe normal precautions appropriate to the circumstances and quantity of material handled. Eye protection and gloves are recommended.
References
Tuleu C. Paediatric formulations in practice. In: Costello I, Long PF, Wong IK et al. (ed) Paediatr Drug Handl London: Pharmaceutical Press; 2007;43–74.
European Medicines Agency. Committee for Medicinal Products for Human Use (CHMP): Reflection paper: formulations of choice for the paediatric population 2006. Available at: http://www.ema.europa.eu/docs/en_GB/document_library/Scientific_guideline/2009/09/WC500003782.pdf (accessed July 2017)
Fabiano V, Mameli C and Zuccotti GV. Paediatric pharmacology: remember the excipients. Pharmacol Res 2011;63(5):362–365.
Turner MA, Duncan JC, Shah U et al. Risk assessment of neonatal excipient exposure: lessons from food safety and other areas. Adv Drug Deliv Rev 2014;73: 89–101. doi: 10.1016/j.addr.2013.11.003